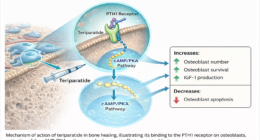
In orthopedic surgery, the difference between successful fracture healing and implant failure can be measured in micrometers. While surgical technique receives considerable attention, the manufacturing precision of implants plays an equally important role in determining patient outcomes. From screw thread accuracy to plate surface finish, manufacturing tolerances directly influence biomechanical performance, osseointegration, and long-term implant survival. Understanding this connection helps distributors, surgeons, and procurement professionals make informed decisions that ultimately benefit patients more.
The Biomechanical Imperative: Why Tolerances Matter
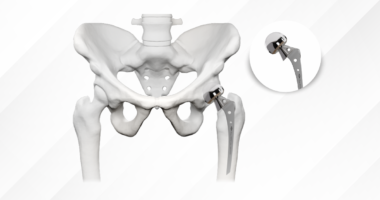
Manufacturing precision determines whether implants perform as biomechanically designed. Locking plate systems provide a clear example: the fixed-angle relationship between screw and plate depends on precise thread geometry and hole dimensions. If manufacturing tolerances drift even slightly, the locking mechanism weakens, the screws toggle, and angular stability disappears. The clinical consequence? Fractures that should heal with stable fixation instead experience micromotion that delays union or causes malunion.
Key areas where precision impacts biomechanics:
- Thread geometry: Improper pitch or depth compromises bone purchase and pull-out strength
- Plate-screw interface: Dimensional variations reduce angular stability and construct rigidity
- Surface finish: Rough surfaces increase soft tissue irritation and inflammatory response
- Hole positioning: Misaligned holes complicate insertion and prevent optimal screw trajectories
Material Consistency and Metallurgical Control
Beyond dimensional accuracy, manufacturing precision extends to material properties and metallurgical consistency. Medical-grade titanium and stainless steel must meet stringent composition standards, but manufacturing processes – heat treatment, cold working, surface passivation – significantly affect final characteristics. Implants from manufacturers with inadequate process control may exhibit variable mechanical properties even within the same production lot, creating unpredictable clinical performance.
Manufacturing factors affecting material performance:
- Heat treatment protocols: Improper thermal processing weakens strength and fatigue resistance
- Surface passivation: Inadequate oxide layer formation increases corrosion susceptibility
- Grain structure: Inconsistent processing creates stress concentration points
- Alloy composition verification: Batch variations affect biocompatibility and mechanical properties
Instrumentation Precision: The Forgotten Variable
Implant performance depends on both the device and the surgical instruments used for insertion. Drill bits, taps, screwdrivers, and targeting guides manufactured to lose tolerances create cascading problems. A worn drill bit creates oversized holes, reducing screw purchase. Imprecise targeting guides cause screws to miss intended trajectories. Even poorly machined screwdriver tips strip screw heads during insertion. Manufacturers committed to precision recognize that quality control must extend to the complete surgical system.
Instrumentation precision requirements:
- Drill bit accuracy: Diameter tolerances within 0.05mm prevent oversized holes
- Targeting guide alignment: Angular precision ensures accurate distal locking
- Screwdriver interface: Exact matching prevents stripping during insertion
- Reusable instrument durability: Maintained accuracy through multiple sterilization cycles
GPC Medical Ltd.: Precision Manufacturing for Global Markets
GPC Medical Ltd. has built its reputation on understanding that manufacturing precision directly translates to clinical reliability. The company’s ISO 13485:2016 certified facilities incorporate CNC machining with automated quality verification, ensuring dimensional tolerances remain within specified ranges. Raw material verification, dimensional inspection using coordinate measuring machines, and surface finish analysis ensure consistent quality across all product lines.
GPC Medical Ltd.’s precision manufacturing advantages:
- ISO 13485:2016 certification: Systematic quality management ensuring consistent processes
- CNC machining capabilities: Computer-controlled production, maintaining tight tolerances
- In-process quality verification: Real-time monitoring, preventing defects
- Comprehensive material testing: Confirming mechanical properties and biocompatibility
- Validated sterilization processes: Ensuring sterility without compromising integrity
Global Trends: Rising Quality Expectations
The global orthopedic implant market increasingly demands manufacturing precision as a baseline expectation. Regulatory frameworks worldwide have intensified, with the European Union’s Medical Device Regulation (MDR) requiring more rigorous clinical evidence. Emerging markets, traditionally more price-sensitive, now increasingly prioritize quality certifications as healthcare systems mature.
Simultaneously, value-based healthcare shifts procurement toward total cost of ownership. Hospital systems recognize that implant complications cost far more than price differentials between precision-manufactured and budget implants, favoring manufacturers like GPC Medical Ltd. that deliver international quality at accessible price points.
Global market trends affecting manufacturing standards:
- Regulatory harmonization: Convergence toward ISO standards raises quality bars
- Traceability requirements: Serialization demands robust documentation
- Emerging market sophistication: Growing quality consciousness globally
- Value-based procurement: Total cost calculations favor quality over price