One of the main issues that patients consider after fracture surgery is whether the orthopedic implants should be removed or not. There is a variety of fixation devices that can either be permanently implanted in the body, where they continue to fulfill their stabilization function and then become inert, or be planned for elimination after healing.
Distinguishing between temporary and permanent fixation is a crucial facet of the patient’s recovery as it enables them to manage their expectations effectively.
Modern orthopedic practice recognizes that implant permanence exists along a spectrum determined by multiple factors: the implant’s design intent, the fracture’s location and healing characteristics, patient age and activity level, and the potential for long-term complications
The Fundamental Distinction: Temporary vs Permanent Fixation
Temporary fixation devices are made with the clear understanding that they will be removed once the bone healing is complete. The most apparent temporary fixation category is external fixation; these scaffolds stabilize fractures via transcutaneous pins or wires connected to external frames, thus providing mechanical support while allowing staged reconstruction and soft tissue management. The temporary nature of these devices does not arise from their material limitations. Still, rather than their functional design, these devices cannot be left in place indefinitely without risks of pin tract infections and patient discomfort.
Internal fixation devices are in a more ambiguous category. In the past, the greater part of plates, screws, and nails were regarded as temporary implants that had to be removed after the fracture healing. Currently, the practice has changed drastically: several internal fixation devices are left in the body permanently unless there are definite reasons for removal. This change reflects improved material biocompatibility, greater understanding of bone-implant interaction, and acknowledgement that removal surgery is a risky procedure.
Key differences between temporary and permanent fixation:
- Design intent: Temporary devices prioritize ease of removal
- Material selection: Temporary uses stainless steel; permanent favors titanium
- Patient counseling: Temporary requires clear removal timing communication
- Cost considerations: Temporary involves multiple procedures
Clinical Indications for Implant Removal
Even when implants are designed for permanent fixation, there are certain clinical conditions that require removal. The most common reason for removal is symptomatic hardware. Patients who have pain, irritation, or functional limitation caused by the implant undergo implant removal after removal once bone healing is mechanical stability. This situation often arises with the plates placed right over the subcutaneous bone surfaces, like the clavicle plates, the distal radius plates, and the tibial plates, where there is barely any soft tissue coverage, and the hardware is palpable.
Pediatric patients are a special group where implant removal matters more. The growing bones change a lot through remodeling, and the implants extending through the growth plates may create physeal bars, resulting in growth disturbances or angular deformities. Besides, the long lifespan expected for pediatric patients gives rise to concerns about the accumulation of metal ions and the chance of implant failure at a later time. Therefore, the orthopedic surgeons very often advise the removal of the hardware in children after the bone has healed completely.
Common indications for implant removal:
- Symptomatic hardware: Pain or irritation causing patient discomfort
- Infection: Hardware removal is necessary to eradicate the infection
- Hardware failure: Broken plates or screws requiring removal
- Pediatric considerations: Growth plate protection and metal exposure concerns
- Planned staged procedures: Temporary fixation removed after soft tissue recovery
When Implants Remain Permanently: The Modern Standard
Contemporary orthopedic practice is generally moving towards permanent retention of implants unless clinical factors justify removal. The reason for this change is that, over time, the evidence has shown that regularly removing asymptomatic, well-positioned implants does not really benefit patients but rather exposes them to the risks of surgery, such as complications of anesthesia, wound infections, and refracture during the post-removal period when bone remodeling leads to temporary mechanical weakness.
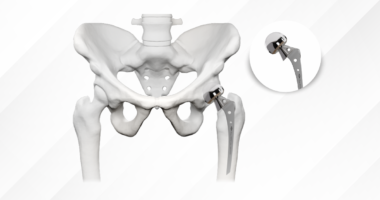
Intramedullary nails are a good example of implants that are usually kept permanently. Since they are located inside the medullary canal and thus separated from the subcutaneous surfaces, nails rarely cause symptoms that would necessitate their removal. Besides, nail removal is a procedure that requires special tools, and it is quite risky as it may cause an intraoperative fracture. Likewise, plates that are well fixed in anatomical locations that are covered by soft tissues to a large extent are normally left permanently unless there is a development of specific problems.
Factors favoring permanent implant retention:
- Asymptomatic status: Absence of pain or functional limitation
- Deep anatomical positioning: Implants beneath soft tissue rarely cause symptoms
- Elderly patients: Limited remaining lifespan reduces long-term concerns
- Titanium composition: Superior biocompatibility supports safe retention
- Risk-benefit analysis: Removal surgery risks exceed theoretical complications
GPC Medical Ltd.: Comprehensive Solutions for Temporary and Permanent Fixation
GPC Medical Ltd. has created a wide range of orthopedic trauma products capable of supporting all fixing situations. With more than forty years of manufacturing experience and presence in more than 150 countries, the company supplies comprehensive systems that assist surgeons throughout the whole treatment continuum.
The external fixation systems of the company represent the ideal temporary fixation design. The range of GPC includes single-sided frames for quick damage control, circular fixators for complex deformity correction, and hybrid systems that combine external stability with internal fixation. These modular systems allow intraoperative revision and staged reconstruction, while the sterilizable instruments keep the per-case costs low and at the same time provide accuracy.
GPC Medical Ltd.’s temporary fixation solutions:
- exHEAL Monorail Limb Reconstruction System
- exHEAL Ilizarov Ring Fixation System
- exHEAL HF2 External Fixation System
- exHEAL Dynamic External Fixation System
- exHEAL Circular/Round Clamp External Fixation System
- exHEAL AO Type Tubular External Fixator System
For permanent internal fixation, GPC manufactures a complete range of plates, screws, and intramedullary nails with the material properties and precision that long-term retention demands. The company’s locking plate systems utilize medical-grade titanium alloys, providing superior biocompatibility and promoting osseointegration. Anatomically contoured designs minimize soft tissue irritation.
GPC’s intramedullary nailing systems are engineered for permanent retention with cannulated designs permitting guidewire-assisted insertion and interlocking screw systems providing rotational stability. The company’s ISO 13485:2016 certification, CE Mark, and FDA registrations ensure manufacturing precision translates to predictable clinical performance.
GPC Medical Ltd.’s permanent fixation portfolio:
- fixLOCK Plating System
- intraHEAL Nailing System
- vaLOCK Plating System
- Distal Radius System
- Non-Locking Plating System
- Cannulated Screw System
- spineHEAL Complete Spine System
Making Informed Decisions: A Collaborative Process
The decision to remove or retain orthopedic implants should emerge from collaborative discussion between surgeon and patient, weighing clinical factors against individual circumstances. Patients benefit from understanding that implant permanence is not predetermined but represents an ongoing assessment: asymptomatic hardware generally remains, while symptomatic implants or specific clinical indications warrant removal.
Manufacturers like GPC Medical Ltd. support these decisions by providing implant systems engineered for their intended purpose—temporary devices designed for safe removal, permanent implants optimized for long-term biocompatibility, and comprehensive product lines giving surgeons flexibility across treatment scenarios. Understanding when implants are removed transforms from an abstract question into a personalized discussion accounting for fracture healing, implant position, patient age, symptom presence, and the balance between removal risks and retention benefits.